A second chance for the NHS to use early Alzheimer’s drugs?
The National Institute for Health and Care Excellence (NICE) will review, for a second time, NHS use of the early Alzheimer's drugs, lecanemab and donanemab. The breakthrough drugs, which are the first in the world to be disease-modifying, were rejected for NHS patient use due to the high cost, in 2025.
However, they have been licensed for private use since 2024, this means that for patients who can afford to pay for them can receive them in a private hospital or private health care setting.
How do the Alzheimer’s drugs work?
In Alzheimer’s disease there is a build up of amyloid proteins which causes damage to the brain and brain cells.
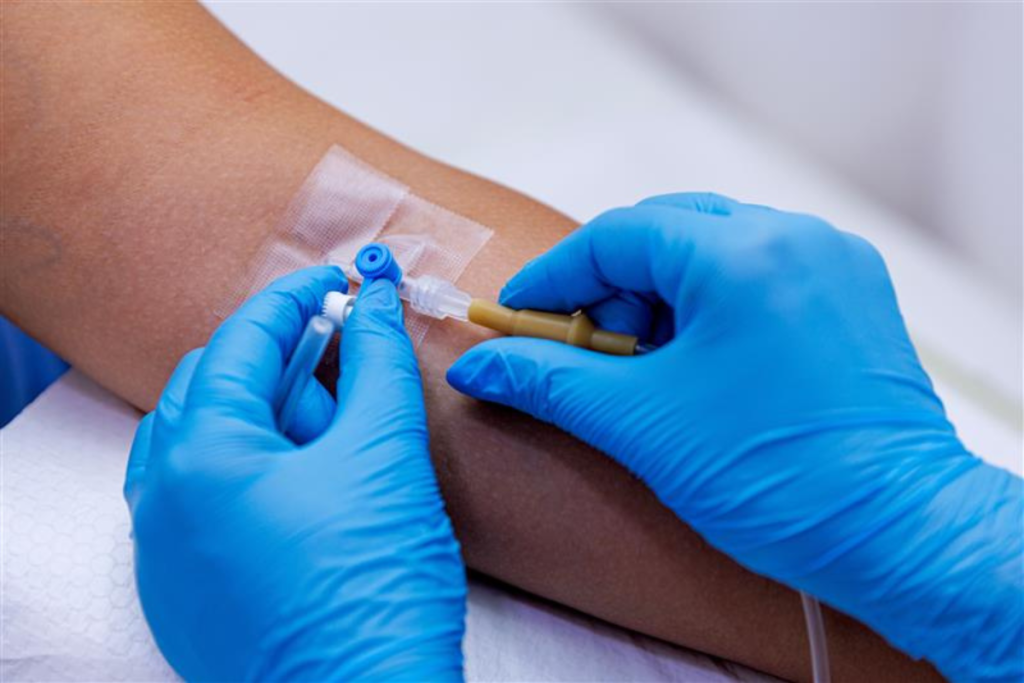
Lecanemab and donanemab work by removing the protein build up from the brain. The drugs are given intravenously, through the arm, and this can drug can only be offered in a medical setting.
The drugs are disease-modifying, meaning they can slow down the condition for several months and improve the quality of life for the person living with Alzheimer’s. These drugs are not a cure.
Who can have the drugs?
In the UK, only people with a diagnosis of mild or early-stage Alzheimer’s disease or people with Mild Cognitive Impairment, who have high levels are amyloids, are eligible.
Some people living with Alzheimer’s, in the UK, have accessed the drugs by volunteering in clinical trials and others have paid privately for the drug.
What are the benefits?
Research shows that memory and thinking issues are reduced by around 27%-35% for those in the early stages of Alzheimer’s/MCI.
BRACE CEO, Liberty Harrison shares:
“News that families affected by Alzheimer's may get a second chance at accessing early treatments on the NHS is very welcome. Alzheimer's is the most common form of dementia in the UK, and its cost to patients, families and society is far too high. Wider access to these disease-modifying treatments could give thousands of people a better quality of life for longer.”
Share this page
